Discovery of Natural Product
Hyaluronidase Inhibitors
Production of Extracts
1
Screening for Hyaluronidase Inhibitory activity
2
Toxicity Testing
3
Testing in Cell Models
4
Formulation & Testing
5
Testing in Humans & Animals
6
Select Botanicals:
2500

Create Extracts:
5000

Screen for Activity:
250
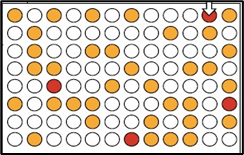
Test in Cell Models:
10
Control:
+ Inhibitor:
Confirm Findings:
3

Current Applications
At Epidarus, we are taking a different and potentially complementary approach to restoring healthy levels of HMW-HA throughout the body. Our bio-active Hyaluronidase Inhibitors work to slow the breakdown of HA, thereby increasing the amount of HMW-HA, while also lowering the levels of immuno-stimulatory HA fragments.
Osteoarthritis
In joints, HA has two distinct and complementary functions, and degradation of HA is a major driving force in the progression of Osteoarthritis.
- Cartilage contains HA which is linked to a number of specialized proteins to form a compressible, shock-absorbing cushion between bones. Breakdown of HA results, ultimately, in bone rubbing on bone during movement and this is very painful. The HA fragments are also powerfully immunogenic and contribute to the painful inflammation seen in arthritic joints
- Synovial fluid is essentially a strong (> 10 mg/ml) solution of HMW-HA. The lubricating or gliding properties of synovial fluid which bathes joints are dependent on the water-binding ability of intact HA molecules. This lubrication is lost when the HA is degraded, further adding to the stress on affected joints
Only one drug is currently approved to treat Osteoarthritis by targeting the underlying biological processes that are causing cartilage destruction. This drug, Pentosan Polysulfate, acts by inhibiting Hyaluronidase-mediated HMW-HA degradation and is effective in treating Osteoarthritis in horses and dogs. However, side-effects limit the use of this drug, and it is not approved in the US to treat Osteoarthritis in either humans or animals.
In our laboratory models of articular cartilage and synovial fluid, our Hyaluronidase inhibitors effectively prevent HMW-HA degradation and are powerfully anti-inflammatory. The effects we see are comparable to or greater than Pentosan Polysulfate in all experiments.
We are currently setting up a trial in naturally occurring Osteoarthritis in pet dogs. This trial will be conducted in Europe, where we can directly compare the effects of Epidarus extracts with those of Pentosan Polysulfate.
BLADDER INTERSTITIAL CYSTITIS
While researching the bioavailability of our extracts, we found that the Hyal-1 inhibition was occurring in bladder after oral dosing. This led us recognize the potential use of our extracts to treat Bladder Interstitial Cystitis (BIC), a chronic and extremely painful condition for which treatment options are currently very limited. BIC occurs without any bacterial infection and is thought to result from over-stimulation of the patient’s immune cells, particularly Mast Cells.
HA is an extremely important molecule in the bladder, where is forms a protective coating that shields the bladder from the toxic irritants in urine. The chronic, painful inflammation in BIC is associated with an accumulation of inflammatory HA fragments which hyper-stimulate the immune system and set up a vicious cycle of immune-mediated destruction of the protective HA layer.
Pentosan Polysulfate, Elmiron®, is the current standard drug used to treat BIC and is approved for this purpose worldwide, including the US. However, its poor oral bioavailability means that high doses must be given and serious side-effects, including bleeding and vision damage are strong contra-indications for its use.
Our lab tests have shown that Epidarus extracts can increase the amount of HMW-HA in cultured bladder epithelial cells far more effectively than Pentosan Polysulfate and we are starting to test oral formulations of the extracts in patients. Our initial patient data are very encouraging, and we are confident that inhibiting the breakdown of HMW-HA in the bladder will be a major component of future treatment regimens for this condition.
ROSACEA
While Rosacea is a multi-factorial disease, it is clear from many studies that chronic hyper-activation of the immune system plays a central role in the pathophysiology of the disease. Since immune modulation by HA fragments drives many such conditions, we are investigating the role of HA metabolism in Rosacea. Based on the literature in this field which shows a strong connection between Mast Cells activation and Hyaluronidase release, we have looked at the effects of our botanical extracts on Mast Cells which we have isolated from human facial skin. We found that our Hyaluronidase inhibitors very effectively inhibit granule release -- degranulation -- and decrease histamine production by more than 90%. Mast Cells initiate and amplify inflammatory responses and are abnormally activated in the skin of Rosacea patients.
Based on these results, we have been testing cream formulations of our botanical extracts in patients suffering from Rosacea.
Before:
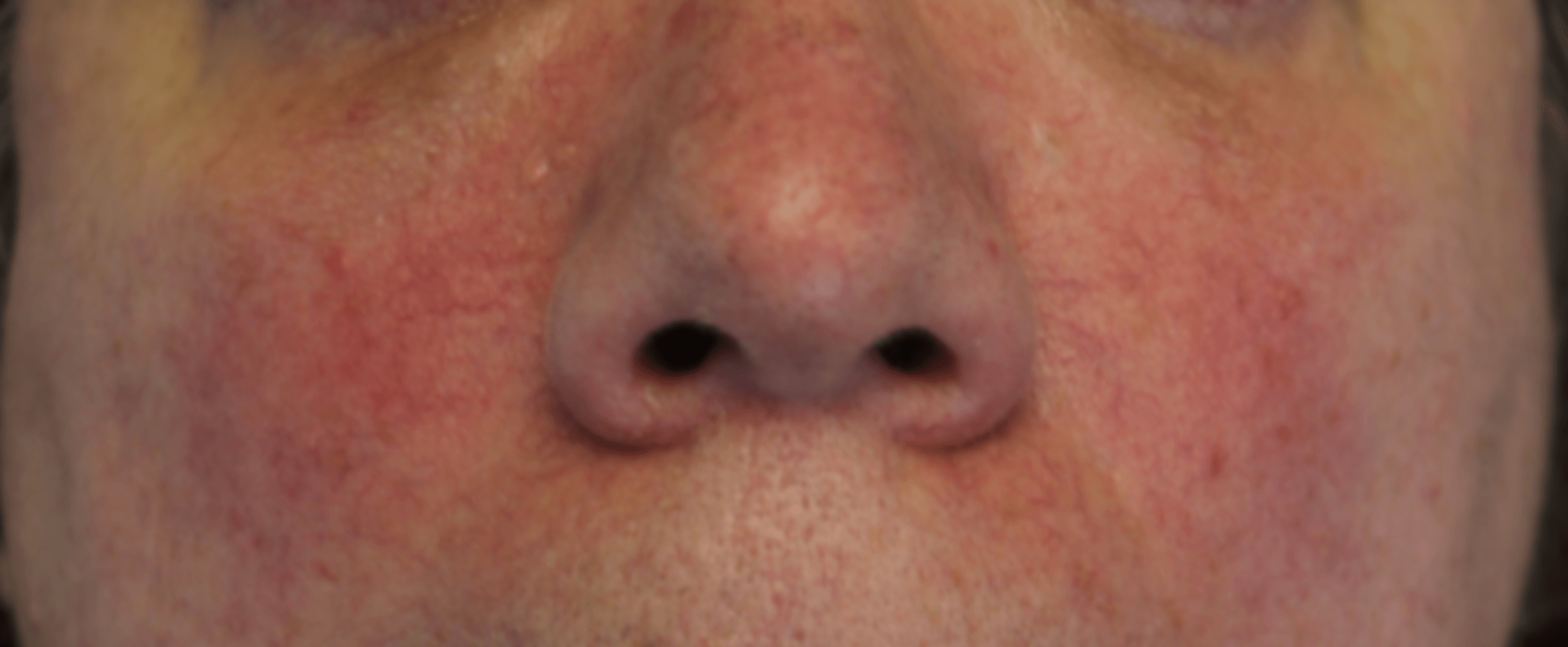

After:
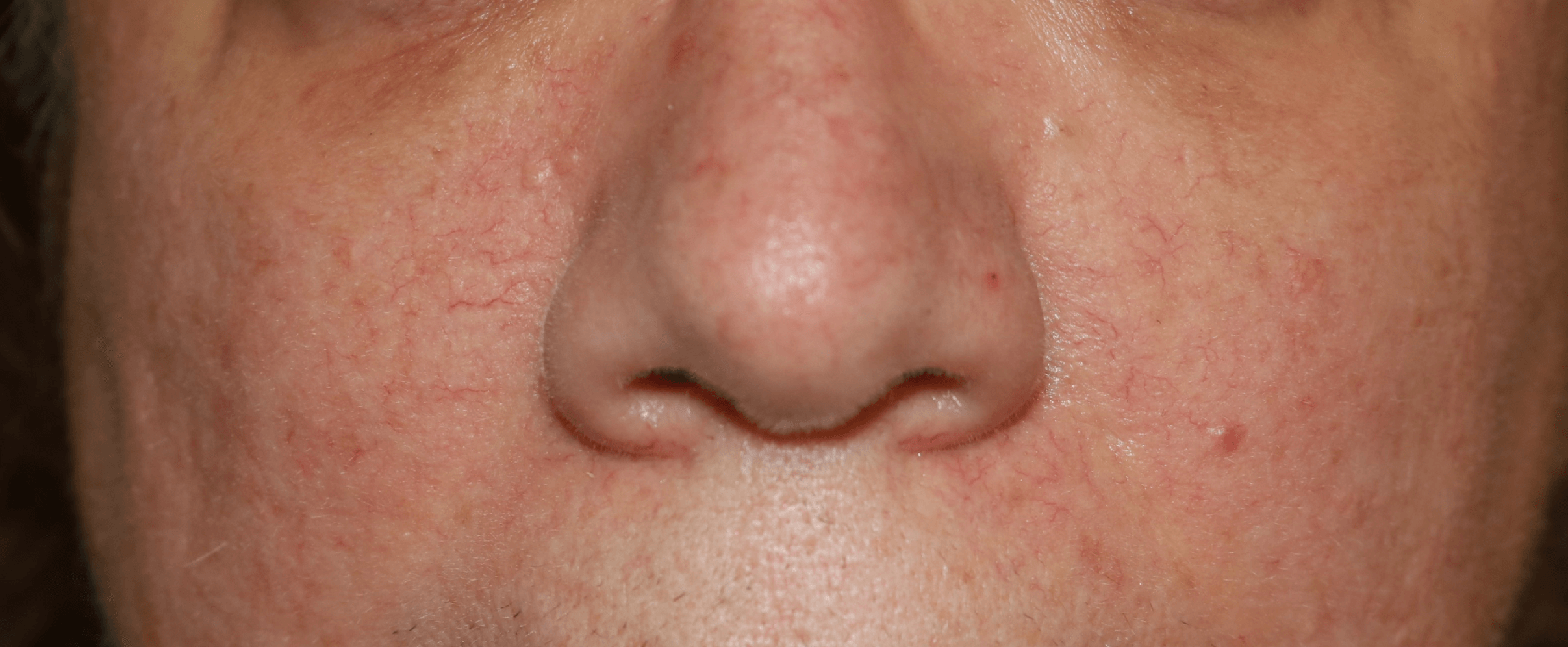
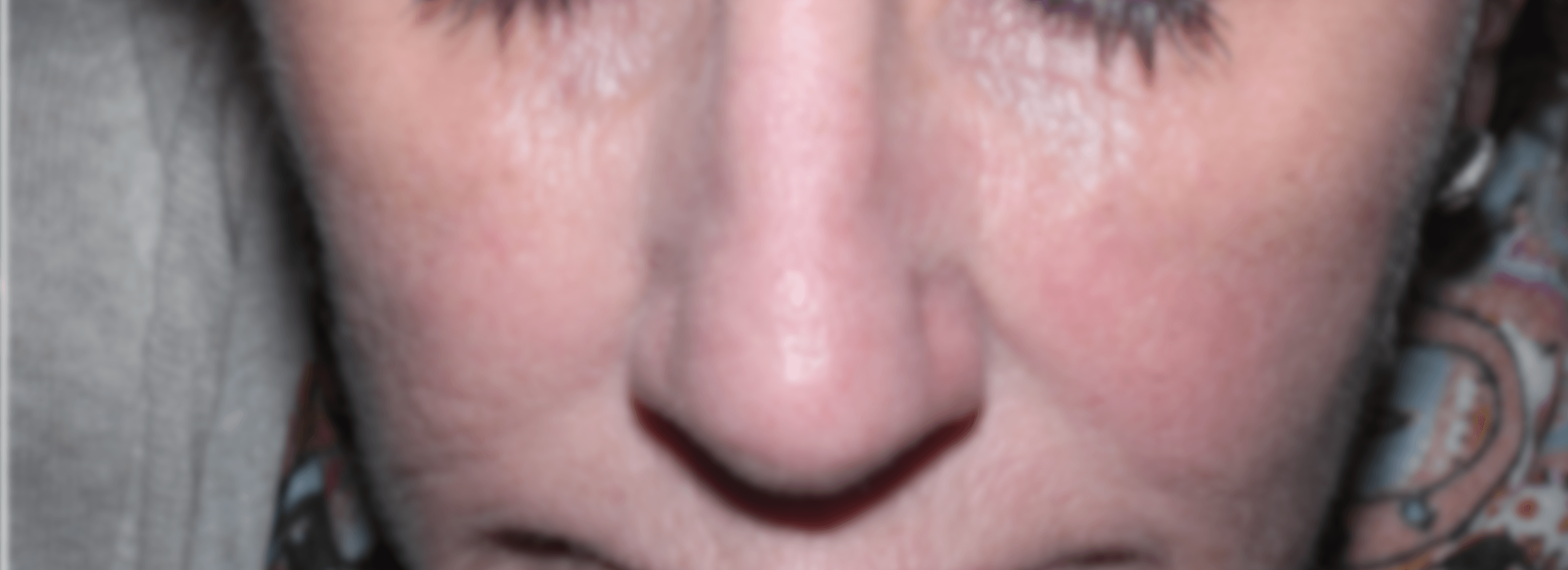
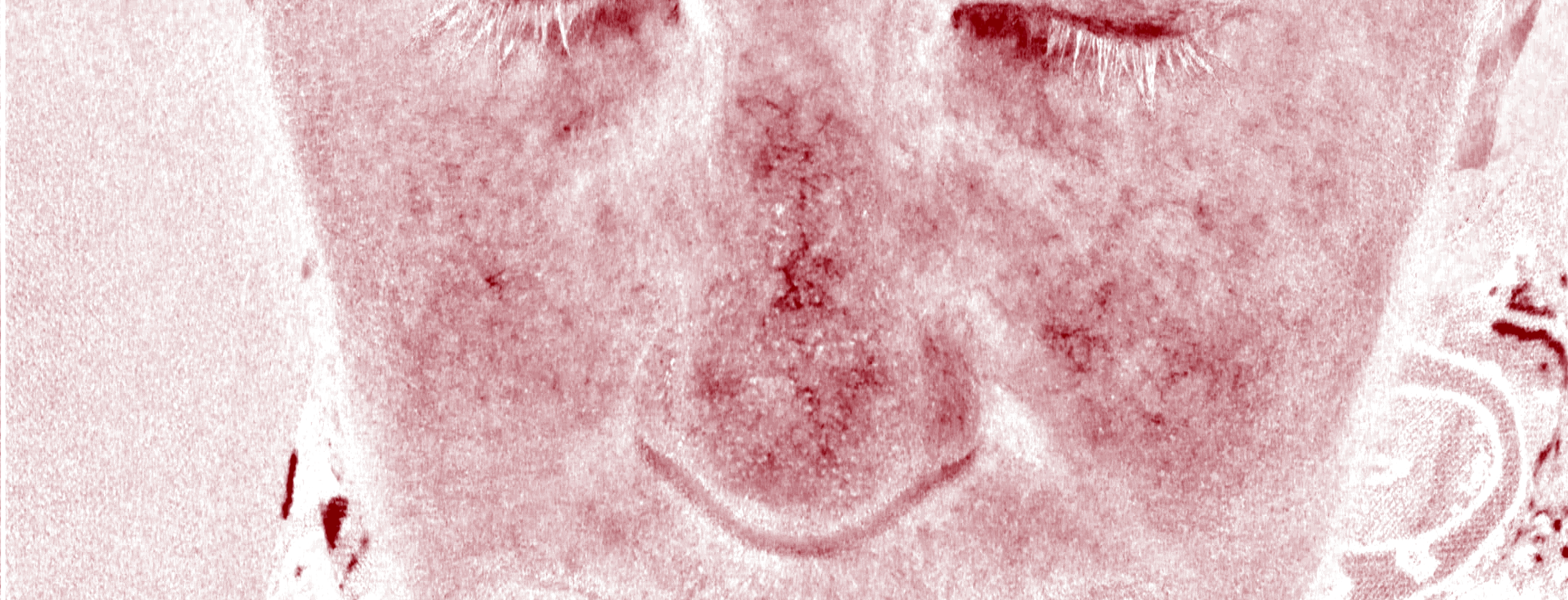
Patients enrolled in the study are photographed at 3-week intervals using standard photography and a Reveal® camera to visualize redness and small blood vessels
Modifying HA Levels for Healthier skin
As skin ages, the amount of HMW-HA decreases and the lower molecular weight fragments increase. Since one of the primary functions of HA in skin is to bind and retain moisture, aged skin is dry and fragile. HA also protects the skin from damaging UV rays in sun and loss of HMW-HA means that older skin is much less well protected against sun damage.
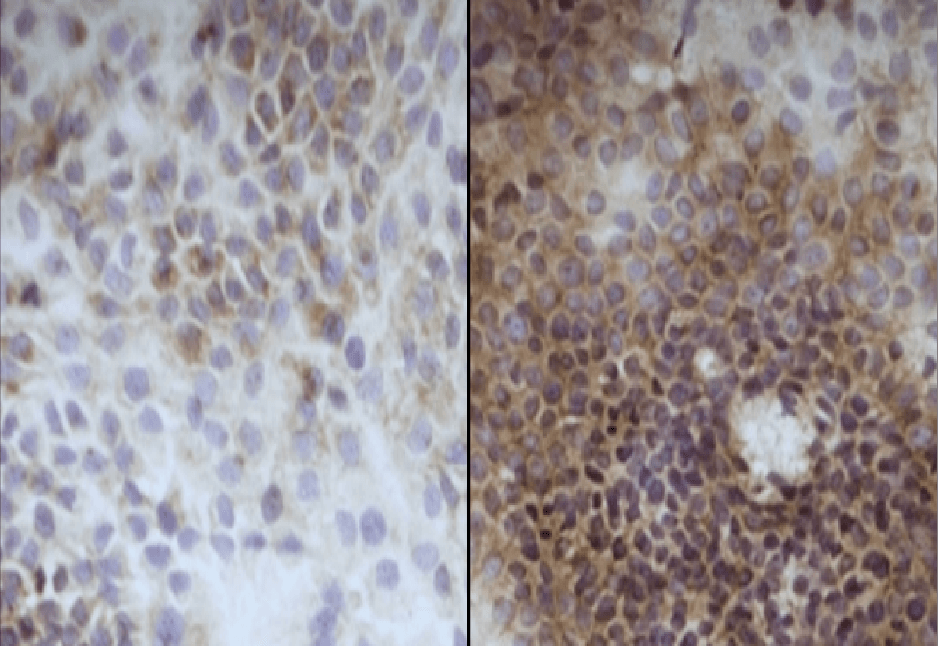
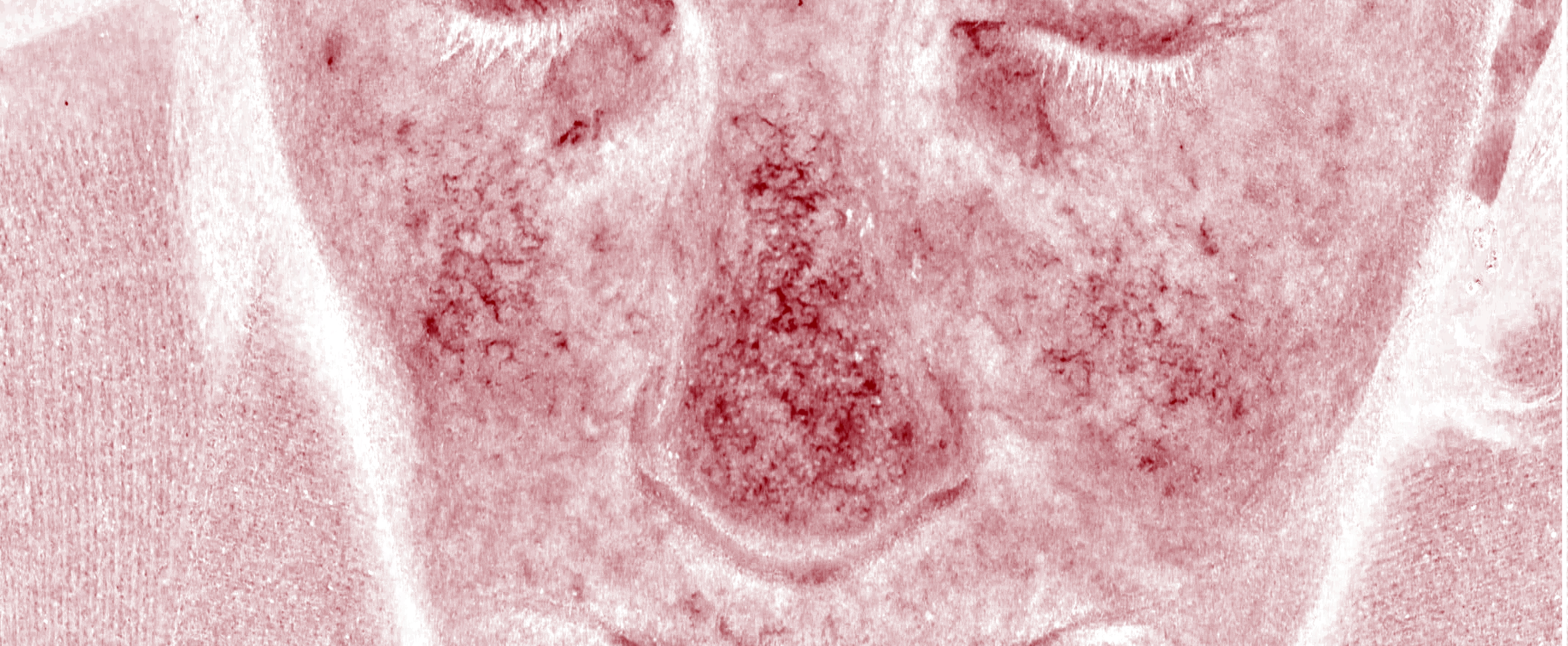
We have investigated the effects of Epidarus extracts using human skin taken from patients who are undergoing facial plastic surgery. This allows us to look at the potential beneficial effects in appropriately aged skin.
Obtain Patient Skin

Prep Skin for Testing
Control
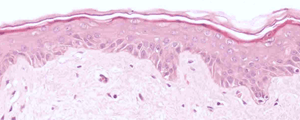
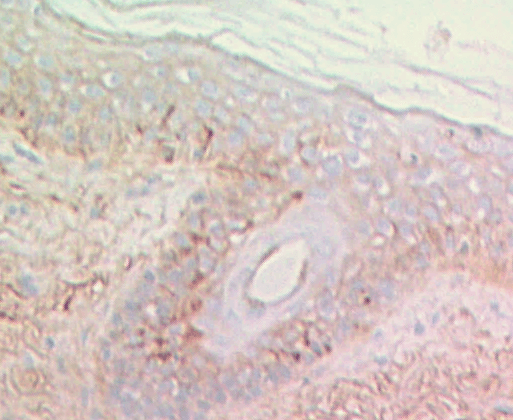
H&E
Stain
HA
Stain
+Extracts
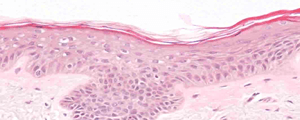
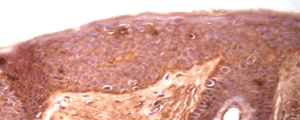
Stain Microscope Sections to See Cell layers & HA
Each day, in a healthy individual, about 15 grams of HA is synthesized and approximately 50% of it is broken down in a tightly regulated fashion. In skin, the half-life of HA is approximately 1 day, so an intervention which allows intact HA to accumulate effectively enables the skin to be moisturized from within and to support stronger, healthier skin.